Back to Knowledge Base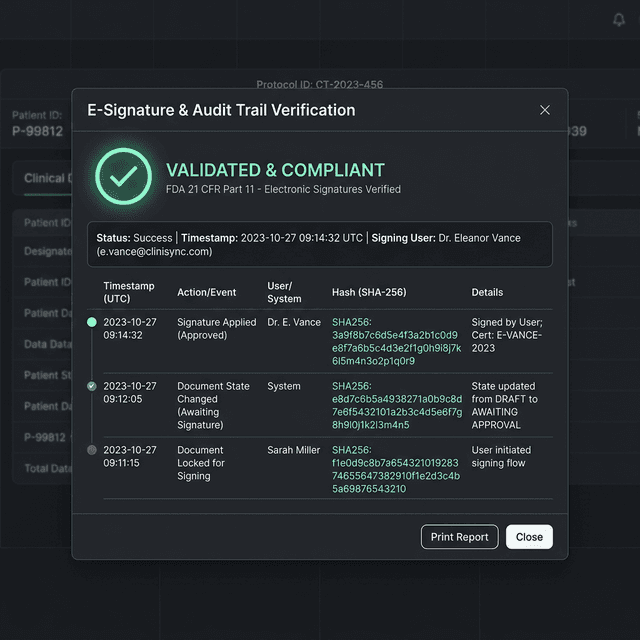
FDA CFR 21 Part 11 Primer
An overview of how TARE helps you achieve compliance with FDA Part 11 electronic records regulations.
Compliance isn't just a feature; it's engineered into the core of TARE. We built our database architecture specifically to meet the stringent requirements of FDA 21 CFR Part 11, securing electronic records and electronic signatures.
Core Compliance Pillars
Immutable Audit Trails
Every action (create, read, update, delete) is permanently logged at the database trigger level. Changes to records automatically generate a non-erasable delta log of the old and new values.
Electronic Signatures
When a protocol or dataset is finalized, it can be electronically signed. The signature is cryptographically bound to the specific revision of the data using SHA-256 hashes.

Configuring Approvals
- Navigate to Settings > Compliance Rules.
- Toggle Require Counter-Signatures if your workflow needs a secondary reviewer.
- Set up Authentication Timeout periods to force re-authentication during critical signing events.
